Introduction
The rare earth elements, particularly the lanthanides, have made their mark in modern science. Among these, lutetium stands out for its unique combination of chemical properties, favourable isotope decay, and physical properties, having made a revolutionary impact in specialised materials, medical imaging and therapy.1–3 Because of its scarcity, very few experiments have been performed to fully investigate its potential in a multitude of applications. In the last few decades, lutetium has played a central role in the development of advanced materials, high-performance electronics, and, most notably, targeted cancer therapies.4–8 Additionally, the recent clinical adoption of lutetium-177 radiopharmaceuticals has placed lutetium at the forefront of nuclear medicine, offering new hope for patients with previously intractable malignancies.9 This article discusses lutetium, both naturally-occurring and radioactive, from its discovery to its ever-expanding clinical and technological applications.
Discovery and chemical behaviour of lutetium
In 1907, French chemist Georges Urbain and Austrian mineralogist Baron Carl Auer von Welsbach (Fig. 1) independently isolated lutetium from the mineral ytterbia, a oxide crystal containing ytterbium oxide and lutetium oxide.10 Urbain named the element after 'Lutetia', the ancient Roman designation for Paris, with Welsbach proposing the name cassiopeium.10 Ultimately, the International Union of Pure and Applied Chemistry (IUPAC) adopted the name ‘lutetium' as a way to credit both researchers for their independent discovery of the element.10 Lutetium has 71 protons, and in its most abundant state, 104 neutrons. Lutetium-176, the only other naturally-occurring isotope of lutetium (2.5% abundance) is radioactive, decaying via beta decay (decay by releasing beta particles like electrons or positrons to be emitted from core) with a half-life of 3.7x1010 years.10,11 Lutetium behaves similarly to other lanthanides while also exhibiting transition metal behaviour, which is due to its filled 4f shell and a single 5d electron. This hybrid character explains much of its chemical versatility.
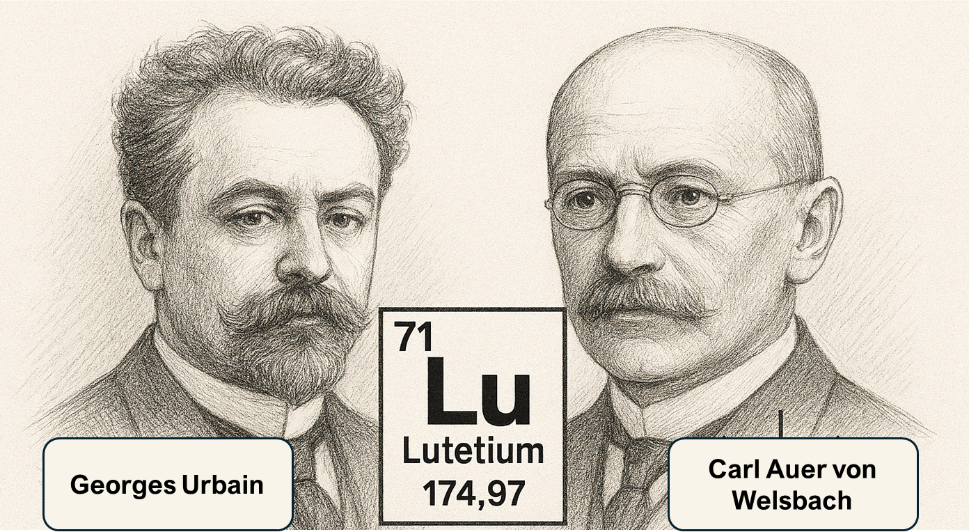
The extraction of lutetium is a highly demanding process, as lutetium shares chemical similarity with other lanthanides.10,12,13 Moreover, it is only found in trace amounts, making it more difficult to obtain in large quantities. Commercially, lutetium is sourced from monazite and bastnäsite, both of which are rare minerals.10 Because of near identical ionic radii and charge of the lanthanides, the use of advanced ion-exchange and solvent extraction techniques is required.13 Additionally, lutetium is also extracted from spallation targets using a similar method outlined by D'yachkov et. al., yielding isotopically pure lutetium isotopes.14 The resultant lutetium exists as either an oxide or salt which can be reduced to metallic form via metallothermic reduction, a process that involves other reactive metals.15
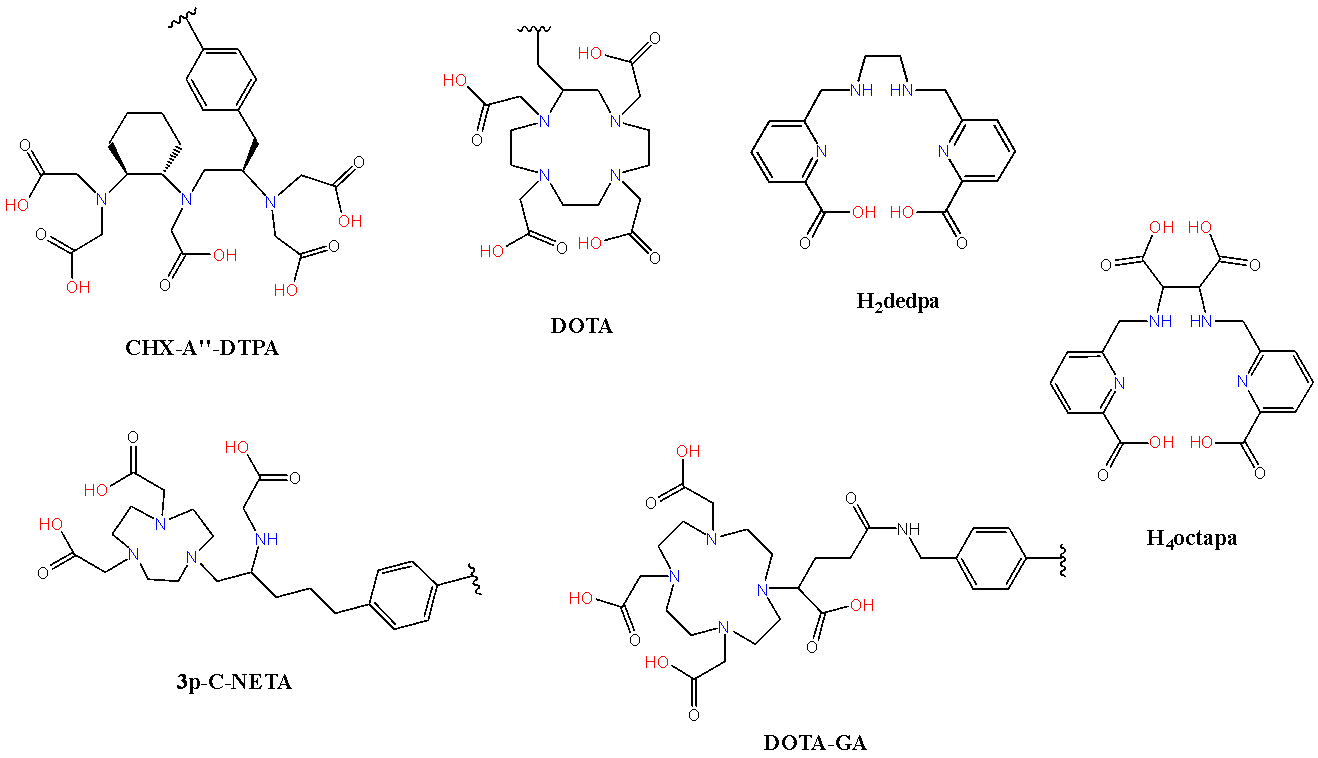
Chemically, lutetium, favours the trivalent oxidation state (Lu³⁺) similar to other lanthanides. Hard and soft acid-base (HSAB) principles are central to understanding lutetium’s coordination chemistry.2 Lu3+ is a strong Lewis acid and therefore has strong affinity for oxygen-donating ligands such as water, phosphates, and carboxylates.2 Lutetium complexes, like other lanthanides, often have high coordination numbers (6–9) and a range of geometries including octahedral, pentagonal bipyramidal, square antiprismatic, and tricapped trigonal prismatic configurations. Because of this, lutetium can complex with a broad range of ligands which are suited to different applications, especially in advanced materials and catalysis.4,8,16 Examples of ligands which coordinate Lu3+ are shown in Fig. 2.17-20
Some applications of lutetium
Enhancing metallic properties
Despite its rarity and high cost, lutetium is occasionally incorporated into high-performance alloys, such as lutetium-zirconia and silver-lutetium alloys, which exhibit exceptional mechanical strength, electrical resistance, corrosion resistance, and high-temperature stability.21–24 These alloys have very small quantities of lutetium, which is enough to impart significant improvements to existing metal properties, resulting in resisting oxidation, and enhancing durability and reliability under extreme conditions.8,22,24 An example of this is Lu/Zr co-doping, where a significant increase in thermal and chemical resistance was observed in SrCeO3 composites for fuel cell development.21 This improvement in physical properties make lutetium-based alloys valuable in various markets such as aerospace, electronics, and specialised scientific instrumentation.
Luminescent materials
Lutetium plays a significant role in luminescent technology and is increasingly becoming more recognised in the field. One of the leading examples of this is lutetium aluminium garnet (LuAG), which is used as a scintillator in radiation detection and as a host matrix for laser phosphors.25 Doping with lutetium, and even other lanthanide activators such as europium or terbium, enables the emission of visible or near-infrared light,4,5,25–27 a property exploited in security screening, analytical instrumentation, and advanced medical imaging. The high density and proton count of LuAG offer superior resistance for high-energy photons, which improves image resolution and brightness in medical imaging devices such as gamma cameras used in positron emission tomography (PET) and single photon emission computed tomography (SPECT) instruments.25 Lutetium oxide (Lu₂O₃) is another lutetium-based material which has been used as dopants in phosphors, as it provides the necessary chemical and thermal stability needed for this application.28 Lutetium can also form complexes with organic frameworks with conjugated π-systems, such as porphyrins and phthalocyanines, that exhibit strong luminescence.4,5 The organic framework absorbs incident energy, transferring it to the lutetium centre for efficient emission, as observed with d-block transition metals. These conjugated systems are utilised and researched in optoelectronics, sensors, and photodynamic therapy, with the flexible coordination chemistry of Lu³⁺ supporting a wide range of ligands leading to different emission profiles.4,5,25,26,28
Superconducting materials
Superconducting materials is a relatively young research field, with many applications still using YBCO (Yttrium Barium Copper Oxide) superconductors.29,30 The exploration of lutetium in superconducting materials is also still in its infancy. Lutetium-containing materials are of particular interest for quantum technology, high-field magnets, and energy-efficient electronics, where low electrical resistance and robust magnetic fields are highly desirable. A concern with superconductivity is the requirement for extremely low temperatures, which limit practical applications. In recent years, lutetium hydride-based superconductors which exhibit superconductivity at temperatures closer to 273 K have been discovered.31–33 The limited supply and extraction cost of lutetium limit its application in various fields. However, research continues to address supply and optimisation in broader applications.
Radiopharmacy: the emergence of lutetium-177
Lutetium-177 (177Lu), a radioactive isotope of lutetium, is important in the medical field in targeted radionuclide therapy.9,34–36 177Lu has a surplus of neutrons and decays via beta emission, emitting a medium energy electron which can deposit that energy onto a tumour cell, causing single strand DNA damage.37 Beta decay also deposits this energy beyond a single cell, and up to 2 mm from the source, making it a perfect therapy for small tumour treatment.38 Because the beta decay energy of 177Lu is low (Eavg. = 134 keV), minimal irradiation damage occurs to surrounding, healthy tissues, further making 177Lu a suitable radioisotope for cancer treatment.39,40 The half-life of 177Lu of 6.65 days ensures that an appropriate dosimetry is delivered during treatment to eradicate cancer cells. Additionally, 177Lu decays with suitable gamma ray energy [Eγ = 208 keV (10%)], which lends itself to low-energy SPECT imaging.41,42 This can be performed along with the treatment administered - a complete theragnostic profile (therapy and diagnostic) (Fig. 3).
Unlike with most applications, radiopharmaceuticals are administered in very small quantities (femto- to nanomole range). Supply is therefore often not a concern. 177Lu is produced in two ways37:
- Direct method via neutron bombardment of 176Lu. A single neutron is added to the nucleus of 176Lu, which converts to 177Lu. This approach yields both 177Lu and 177mLu, a metastable radioisotope which has a half-life of 160 days, which is not favourable for clinical use. Additionally, only isotopically-pure 176Lu can be used, which is in very low abundance, making this approach very difficult to implement in mass production.
- Indirect method via neutron bombardment of 176Yb. A single neutron is added to the nucleus of 176Yb (12.9% abundance), which converts to 177Yb. 177Yb decays via neutron emission, with a half-life of 1 hour, to yield 177Lu. In practice, the target (after bombardment) is left for a minimum of 24 hours to allow > 99% of 177Yb to decay to 177Lu. This is the current method used to produce 177Lu.
Targeted radionuclide therapy (TRNT) using lutetium-177 conjugated to PSMA-617 (a pseudopeptide which binds to a prostate cancer biomarker called prostate specific membrane antigen, colloquially shortened to PSMA) has shown promise as a treatment for metastatic castration-resistant prostate cancer (mCRPC), particularly in patients who have undergone chemotherapy and received androgen receptor pathway inhibitors.44–47 [177Lu]Lu-PSMA-617 is favoured for its optimal half-life, cost-effectiveness, and beta emission, leading to its approval by the Food and Drug Administration (FDA) and the European Medicines Agency (EMA).46 Additionally, diagnostic variants (like gallium-68) of the same molecule are also available, making progression of the treatment much easier to follow.48 Clinical trials have demonstrated efficacy and safety, but a notable number of patients have no response to the treatment or experience disease progression after initial benefits.47,49–51 Alternative treatments are limited for these patients but this will soon change with an increase in clinical trials replacing 177Lu with newer, more potent radionuclides.
Neuroendocrine tumours (NETs) refer to a group of cancers that can arise in various organs such as the lung, gastrointestinal tract, or pancreas.52 Another lutetium-177 TRNT agent that has been approved by the FDA and EMA is [177Lu]Lu-DOTA-TATE, a somatostatin targeting agent which is currently used to treat advanced midgut neuroendocrine tumours.53 Many different analogues of the octreotide (targeting moiety) exist, and these radiolabelled analogues have become an established second- or third-line treatment option for patients with progressive well-differentiated (grade 1–2) gastroenteropancreatic (GEP) neuroendocrine tumours (NETs).52,54–56 Due to the increasing incidence and prevalence of GEP-NETs over recent decades and the development of NET-specific treatments and treatment protocols, there is a growing utilisation of such systemic treatment.52,57
Future of lutetium-177
Lu-177 has been at the forefront of TRNT for the last decade. Since the approval of the PSMA and GEP-NET agents, many patients have successfully undergone treatment for prostate and neuroendocrine cancers.53,58 Despite the clinical success of these two agents, there are still many patients that do not respond to the therapy.7,46,58–60 This could be due to the mechanism of action with beta emission, as single-strand DNA breaks are repairable and could result in cancer cells not being successfully eradicated.61 In contrast, alpha (He2+) and cascading electron (Auger and conversion electrons) radionuclides deposit high energy over a short distance (a few cell lengths), resulting in irreparable double-strand DNA damage.61 Currently, terbium-161 and actinium-225 are being investigated for their efficacy in patients who do not respond to Lu-177, with promising results.49,51,62 Despite the emergence of alternative radioisotopes, the established clinical efficacy of Lu-177, in conjunction with its well-characterised safety profile and robust supply chain infrastructure ensure its continued prominence in targeted radionuclide therapy. The reliability of GMP-compliant production and the integration of lutetium-177 with diagnostic precision provide a strong foundation for future expansion.
Conclusions
The use of lutetium has evolved to become a cornerstone of modern technology and medicine. Its unique chemical properties underpin advances in materials and medical science. With ongoing research into new applications, production methods and complementary radioisotopes, lutetium’s contribution to science is set to expand further. Continued interdisciplinary collaboration between chemists, physicists, clinicians and engineers will be essential to fully realise the therapeutic and technological potential of this remarkable element.
References
- Mishiro, K.; Hanaoka, H.; Yamaguchi, A.; Ogawa, K. Coordination Chemistry Reviews 2019, 383, 104–131.
- Weller, M.; Rourke, J.; Armstrong, F.; Lancaster, S.; Overton, T. Inorganic Chemistry, 7th ed.; Oxford University Press, 2025.
- Kostelnik, T. I.; Orvig, C. Chemical Reviews 2019, 119 (2), 902–956.
- Nosov, V. G.; Kupryakov, A. S.; Kolesnikov, I. E.; Vidyakina, A. A.; Tumkin, I. I.; Kolesnik, S. S.; Ryazantsev, M. N.; Bogachev, N. A.; Skripkin, M. Y.; Mereshchenko, A. S. Molecules 2022, 27 (18), 1–11.
- Tsvirko, M. P.; Kalota, B.; Mikus, A.; Ostrowski, S. Journal of Applied Spectroscopy 2020, 87 (5), 789–795.
- Dash, A.; Chakraborty, S.; Pillai, M. R. A.; Knapp, F. F. R. Cancer Biotherapy and Radiopharmaceuticals 2015, 30 (2), 47–71.
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P. L.; Kulke, M. H.; Jacene, H.; Bushnell, D.; O’Dorisio, T. M.; Baum, R. P.; Kulkarni, H. R.; Caplin, M.; Lebtahi, R.; Hobday, T.; Delpassand, E.; Van Cutsem, E.; Benson, A.; Srirajaskanthan, R.; Pavel, M.; Mora, J.; Berlin, J.; Grande, E.; Reed, N.; Seregni, E.; Öberg, K.; Lopera Sierra, M.; Santoro, P.; Thevenet, T.; Erion, J. L.; Ruszniewski, P.; Kwekkeboom, D.; Krenning, E. New England Journal of Medicine 2017, 376 (2), 125–135.
- Gschneidner, K. A.; Calderwood, F. W. Bulletin of Alloy Phase Diagrams 1988, 9 (6), 690–691.
- Sartor, O.; Herrmann, K. Journal of Nuclear Medicine 2022, 63 (6), 823–829.
- Gross, J. L.; Thoennessen, M. Atomic Data and Nuclear Data Tables 2012, 98 (5), 983–1002.
- Bhardwaj, R.; Ponsard, B.; Sarilar, M.; Wolterbeek, B.; Denkova, A.; Serra-Crespo, P. Applied Radiation and Isotopes 2020, 156, 108986-108992.
- Trtić‐Petrović, T. M.; Kumrić, K. R.; Đorđević, J. S.; Vladisavljević, G. T. Journal of Separation Science 2010, 33 (13), 2002–2009.
- Dong, J.; Xu, Y.; Wang, L.; Huang, X.; Long, Z.; Wu, S. Journal of Rare Earths 2016, 34 (3), 300–307.
- D’yachkov, A. B.; Kovalevich, S. K.; Labozin, V. P.; Mironov, S. M.; Panchenko, V. Y.; Firsov, V. A.; Tsvetkov, G. O.; Shatalova, G. G. Quantum Electronics 2012, 42 (10), 953–956.
- Larochelle, T. Rare Earth Element Reduction to Metals. In Rare Earth Metals and Minerals Industries; Murty, Y. V, Alvin, M. A., Lifton, J. ., Eds.; Springer International Publishing: Cham, 2024; 257–269.
- Belli, R. G.; Tafuri, V. C.; Garcia, N. A.; Roberts, C. C. Organometallics 2023, 42 (11), 1059–1064.
- Lucio-Martínez, F.; Szilágyi, B.; Uzal-Varela, R.; Pérez-Lourido, P.; Esteban-Gómez, D.; Lepareur, N.; Tircsó, G.; Platas-Iglesias, C. Chemistry - A European Journal 2025, 31 (28), 1–12.
- Price, E. W.; Orvig, C. Chemical Society Reviews 2014, 43 (1), 260–290.
- Price, E. W.; Cawthray, J. F.; Adam, M. J.; Orvig, C. Journal of the Chemical Society. 2014, 43 (19), 7176–7190.
- Cassells, I.; Ahenkorah, S.; Burgoyne, A. R.; Van de Voorde, M.; Deroose, C. M.; Cardinaels, T.; Bormans, G.; Ooms, M.; Cleeren, F. Frontiers in Medicine 2021, 8, 1–12.
- Hu, W.; Chen, W.; Wang, H. International Journal of Electrochemical Science 2020, 15 (4), 3157–3163.
- Gschneidner, K. A.; Calderwood, F. W. Bulletin of Alloy Phase Diagrams 1983, 4 (4), 374–375.
- Li, L.; Yang, H.; Yang, P. ACS Applied Electronic Materials 2022, 4 (12), 6253–6260.
- Zhou, K.; Chen, C.; Liu, Y.; Pang, S.; Hua, N.; Yang, W.; Zhang, T. Intermetallics 2017, 90, 81–89.
- Aghay Kharieky, A.; Rezaee Ebrahim Saraee, K. Solid State Sciences 2020, 108, 106335-106343.
- Sivaranjani, V.; Deepa, P.; Murugesan, P.; Suresh, A. A.; Dhavamurthy, M. Emergent Materials 2025, 8 (6), 3959–4005.
- Camargo, M. A.; Neves, A.; Bortoluzzi, A. J.; Szpoganicz, B.; Fischer, F. L.; Terenzi, H.; Serra, O. A.; Santos, V. G.; Vaz, B. G.; Eberlin, M. N. Inorganic Chemistry 2010, 49 (13), 6013–6025.
- Thoř, T.; Rubešová, K.; Jakeš, V.; Cajzl, J.; Nádherný, L.; Mikolášová, D.; Kučerková, R.; Nikl, M. Journal of Luminescence 2019, 215,116647-116654.
- Zhang, B.; Zhang, X.; Zhou, Y.-H. National Science Review 2023, 10 (3), 1–7.
- Pan, Y.; Xin, C.; Wu, W.; Guan, M.; Wang, X. Fusion Engineering and Design 2025, 211, 114802.
- Li, Z.; He, X.; Zhang, C.; Lu, K.; Min, B.; Zhang, J.; Zhang, S.; Zhao, J.; Shi, L.; Peng, Y.; Feng, S.; Deng, Z.; Song, J.; Liu, Q.; Wang, X.; Yu, R.; Wang, L.; Li, Y.; Bass, J. D.; Prakapenka, V.; Chariton, S.; Liu, H.; Jin, C. Science China Physics, Mechanics & Astronomy 2023, 66 (6), 267411.
- Wang, D.; Wang, N.; Zhang, C.; Xia, C.; Guo, W.; Yin, X.; Bu, K.; Nakagawa, T.; Zhang, J.; Gorelli, F.; Dalladay-Simpson, P.; Meier, T.; Lü, X.; Sun, L.; Cheng, J.; Zeng, Q.; Ding, Y.; Mao, H. Matter and Radiation at Extremes 2024, 9 (3), 1-9.
- Zhao, X.; Huang, Y.; Ma, S.; Song, H.; Cao, Y.; Jiang, H.; Huang, Y.; Cui, T. Advanced Science 2025, 12 (3), 1–9.
- Severi, S.; Sansovini, M.; Ianniello, A.; Bodei, L.; Nicolini, S.; Ibrahim, T.; Di Iorio, V.; D’Errico, V.; Caroli, P.; Monti, M.; Paganelli, G. European Journal of Nuclear Medicine and Molecular Imaging 2015, 42 (13), 1955–1963.
- D’Huyvetter, M.; Vincke, C.; Xavier, C.; Aerts, A.; Impens, N.; Baatout, S.; De Raeve, H.; Muyldermans, S.; Caveliers, V.; Devoogdt, N.; Lahoutte, T. Theranostics 2014, 4 (7), 708–720.
- Banerjee, S. R.; Kumar, V.; Lisok, A.; Chen, J.; Minn, I.; Brummet, M.; Boinapally, S.; Cole, M.; Ngen, E.; Wharram, B.; Brayton, C.; Hobbs, R. F.; Pomper, M. G. European Journal of Nuclear Medicine and Molecular Imaging 2019, 46 (12), 2545–2557.
- Dash, A.; Pillai, M. R. A.; Knapp, F. F. Nuclear Medicine and Molecular Imaging 2015, 49 (2), 85–107.
- Poty, S.; Francesconi, L. C.; McDevitt, M. R.; Morris, M. J.; Lewis, J. S. Journal of Nuclear Medicine 2018, 59 (6), 878–884.
- Jowanaridhi, B.; Sriwiang, W. Journal of Physics: Conference Series 2019, 1380 (1).
- Borgna, F.; Barritt, P.; Grundler, P. V.; Talip, Z.; Cohrs, S.; Zeevaart, J. R.; Köster, U.; Schibli, R.; van der Meulen, N. P.; Müller, C. Pharmaceutics 2021, 13 (4), 536.
- Grünberg, J.; Lindenblatt, D.; Dorrer, H.; Cohrs, S.; Zhernosekov, K.; Köster, U.; Türler, A.; Fischer, E.; Schibli, R. European Journal of Nuclear Medicine and Molecular Imaging 2014, 41 (10), 1907–1915.
- Deberle, L. M.; Benešová, M.; Becker, A. E.; Ratz, M.; Guzik, P.; Schibli, R.; Müller, C. Bioconjugate Chemistry 2021, 32 (8), 1617–1628.
- Pathmanandavel, S.; Crumbaker, M.; Ho, B.; Yam, A. O.; Wilson, P.; Niman, R.; Ayers, M.; Sharma, S.; Hickey, A.; Eu, P.; Stockler, M.; Martin, A. J.; Joshua, A. M.; Nguyen, A.; Emmett, L. Journal of Nuclear Medicine 2023, 64 (2), 221–226.
- Banerjee, S. R.; Kumar, V.; Lisok, A.; Chen, J.; Minn, I.; Brummet, M.; Boinapally, S.; Cole, M.; Ngen, E.; Wharram, B.; Brayton, C.; Hobbs, R. F.; Pomper, M. G. European Journal of Nuclear Medicine and Molecular Imaging 2019, 46 (12), 2545–2557.
- Rahbar, K.; Essler, M.; Pabst, K. M.; Eiber, M.; Fougère, C. la; Prasad, V.; Rassek, P.; Hasa, E.; Dittmann, H.; Bundschuh, R. A.; Fendler, W. P.; Kurtinecz, M.; Schmall, A.; Verholen, F.; Sartor, O. Journal of Nuclear Medicine 2023, 64 (4), 574–578.
- Emmett, L.; Willowson, K.; Violet, J.; Shin, J.; Blanksby, A.; Lee, J. Journal of Medical Radiation Sciences 2017, 64 (1), 52–60.
- Sartor, O.; Ledet, E.; Huang, M.; Schwartz, J.; Lieberman, A.; Lewis, B.; Layton, J.; Barata, P.; Jang, A.; Hawkins, M.; Pocha, O.; Lanka, S.; Harris, K. Journal of Nuclear Medicine 2023, 64 (11), 1721–1725.
- Fuscaldi, L. L.; Sobral, D. V.; Durante, A. C. R.; Mendonça, F. F.; Miranda, A. C. C.; da Cunha, M. L.; Malavolta, L.; Mejia, J.; de Barboza, M. F. Pharmaceuticals 2021, 14 (5).
- Sathekge, M.; Bruchertseifer, F.; Vorster, M.; Lawal, I. O.; Knoesen, O.; Mahapane, J.; Davis, C.; Mdlophane, A.; Maes, A.; Mokoala, K.; Mathabe, K.; Van, C.; Wiele, D.; Morgenstern, A. Journal of Nuclear Medicine 2022, 63 (10), 1496–1502.
- Marin, J. F. G.; Nunes, R. F.; Coutinho, A. M.; Zaniboni, E. C.; Costa, L. B.; Barbosa, F. G.; Queiroz, M. A.; Cerri, G. G.; Buchpiguel, C. A. Radiographics 2020, 40 (6), 1715–1740.
- Rosar, F.; Maus, S.; Schaefer-Schuler, A.; Burgard, C.; Khreish, F.; Ezziddin, S. Clinical Nuclear Medicine 2023, 48 (5), 433–434.
- Sowa-Staszczak, A.; Hubalewska-Dydejczyk, A.; Tomaszuk, M. In Recent Results in Cancer Research 2013, 194, 479–485.
- Hennrich, U.; Kopka, K. Pharmaceuticals 2019, 12 (3), 114.
- Kanzaki, T.; Takahashi, Y.; Higuchi, T.; Zhang, X.; Mogi, N.; Suto, T.; Tsushima, Y. Journal of Nuclear Medicine Technology 2020, 48 (4), 326–330.
- Pauwels, E.; Cleeren, F.; Tshibangu, T.; Koole, M.; Serdons, K.; Dekervel, J.; Van Cutsem, E.; Verslype, C.; Van Laere, K.; Bormans, G.; Deroose, C. M. European Journal of Nuclear Medicine and Molecular Imaging 2020, 47 (13), 3033–3046.
- Healy, A.; Ho, E.; Kuo, P.; Zukotynski, K. Frontiers in Nuclear Medicine 2023, 3, 1–6.
- Vermeulen, K.; Vandamme, M.; Bormans, G.; Cleeren, F. Seminars in Nuclear Medicine 2019, 49 (5), 339–356.
- Ladrière, T.; Faudemer, J.; Levigoureux, E.; Peyronnet, D.; Desmonts, C.; Vigne, J. Pharmaceutics 2023, 15 (4),1-22.
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P. L.; Kulke, M. H.; Jacene, H.; Bushnell, D.; O’Dorisio, T. M.; Baum, R. P.; Kulkarni, H. R.; Caplin, M.; Lebtahi, R.; Hobday, T.; Delpassand, E.; Van Cutsem, E.; Benson, A.; Srirajaskanthan, R.; Pavel, M.; Mora, J.; Berlin, J.; Grande, E.; Reed, N.; Seregni, E.; Öberg, K.; Lopera Sierra, M.; Santoro, P.; Thevenet, T.; Erion, J. L.; Ruszniewski, P.; Kwekkeboom, D.; Krenning, E. New England Journal of Medicine 2017, 376 (2), 125–135
- Sgouros, G.; Bodei, L.; McDevitt, M. R.; Nedrow, J. R. Nature Reviews Drug Discovery 2020, 19 (9), 589–608.
- Khazaei Monfared, Y.; Heidari, P.; Klempner, S. J.; Mahmood, U.; Parikh, A. R.; Hong, T. S.; Strickland, M. R.; Esfahani, S. A. Pharmaceutics 2023, 15 (12), 1–26.
- Morgenstern, A.; Apostolidis, C.; Kratochwil, C.; Sathekge, M.; Krolicki, L.; Bruchertseifer, F. Current Radiopharmaceuticals 2018, 11 (3), 200–208.