Abstract
The discovery of ferrocene in the early 1950s revolutionised chemistry. When scientists accidentally created this bright orange solid, its behaviour and chemical properties intrigued the chemistry community, especially due to its unexpected stability in air compared to many other many metal-containing molecules. Its structure, with an iron atom found sandwiched between two hydrocarbon ring-systems, was unlike anything seen before. What began as a curiosity soon became a turning point in chemistry, opening the door to a whole new way of designing molecules. Over time, scientists also discovered that attaching ferrocene to existing drugs could give them new strengths. One of the most promising examples is in cancer treatment. When ferrocene is connected to tamoxifen, a well-known breast-cancer agent, the resulting compound is able to target a wide range of cancers. Ferrocene has also inspired new ideas for treating malaria. By adding it to chloroquine, an older antimalarial drug that no longer works in many regions, scientists created a new drug that can combat drug-resistant parasites. This accidental discovery has grown into a compelling story of innovation, with ferrocene still guiding the search for more effective treatments for some of the world’s most challenging diseases today.
A serendipitous discovery that reshaped chemistry
Nearly seventy-five years ago marked a transformative moment in the evolution of organometallic chemistry with the discovery of bis(η⁵-cyclopentadienyl)iron, widely known as ferrocene (Fig. 1).1 The elegant architecture of ferrocene consisting of an iron atom nestled between two cyclopentadienyl (Cp) rings, resembling a sandwich, was unfamiliar at the time and challenged the established conventions of chemistry and bonding. The electronic arrangement gives ferrocene its stability as well as interesting reactivity, while being quite symmetric in nature.
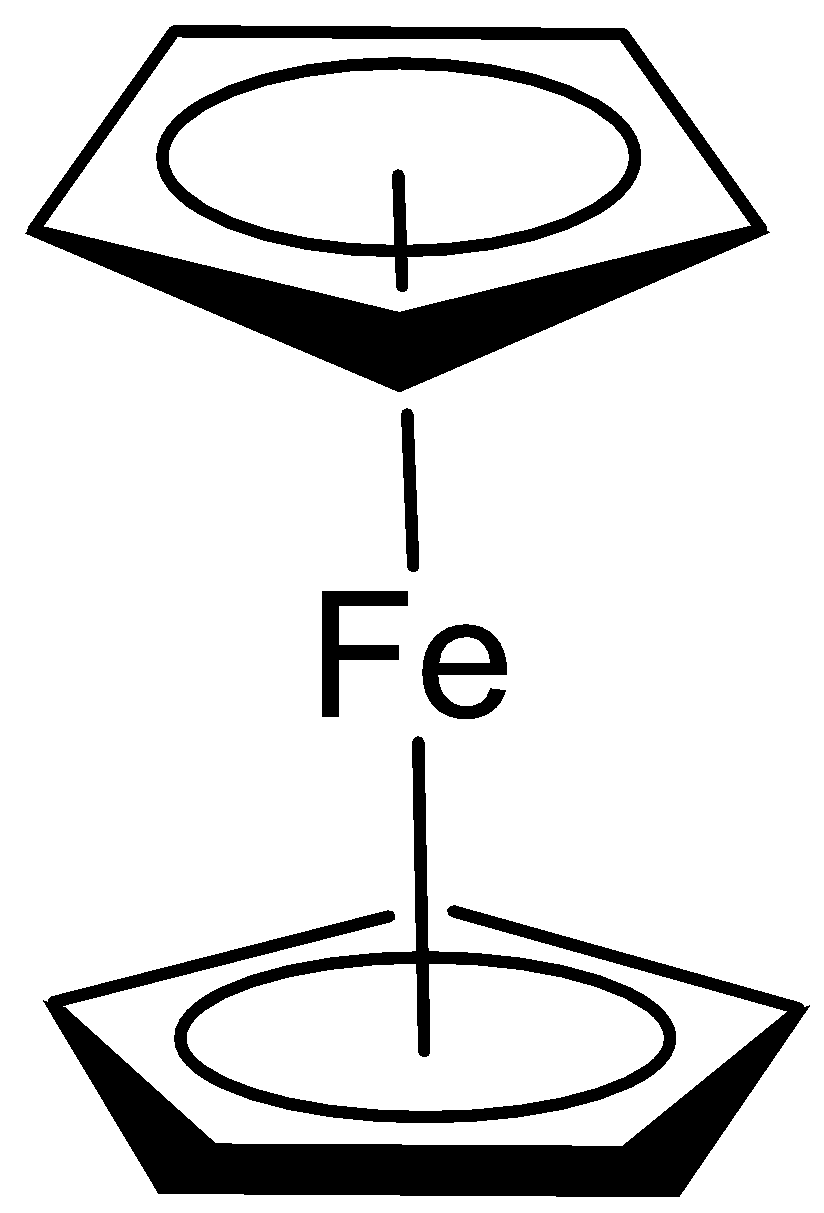

Fig. 1. Bis(η⁵-cyclopentadienyl)iron - ferrocene
Ferrocene was first reported independently by two research groups between 1951 and 1952. Kealy and Pauson attempted the synthesis of fulvalene via the reaction of cyclopentadienylmagnesium bromide with iron(III) chloride at Duquesne University. Instead of isolating fulvalene, the pair isolated an air-stable orange solid. Upon analysis, they determined the molecular formula to be C₁₀H₁₀Fe. Recognising both the novelty and the potential significance of their unexpected discovery, Kealy and Pauson swiftly published their findings in Nature in 1951.2 Unbeknownst to them, a second group from the British Oxygen Company, Ltd consisting of Miller, Tremaine and Tebboth, had synthesised the same compound around the same time. Their approach arose not from targeted synthesis but from research into amine formation using nitrogen and hydrocarbons. In their experiments, a combination of nitrogen and cyclopentadiene was added to an iron catalyst at elevated temperatures and produced a very stable orange substance. Their independent report was published in early 1952,3 contributing to a near-simultaneous recognition of this new and intriguing molecule.
The structure conundrum
Although Pauson and Kealy published the discovery quite rapidly, the structure of the orange compound was not well understood. The pair proposed σ-bonding between the iron atom and carbons of two cyclopentadienyl moieties, with each ring being attached to opposite sides of the metal through a single carbon atom. This proposed structure was instantly questioned and challenged by many including Woodward and Wilkinson at Harvard University, along with Fischer and Pfab at Technische Hochschule München. The main discrepancy between the proposed structure and the data was that the infrared spectrum showed one C-H stretching band, suggesting all C–H bonds to be equivalent, which could not be possible if the metal was bound to a single carbon atom on each ring. Woodward and Wilkinson proposed a revolutionary “sandwich” structure in which the iron atom sat between two cyclopentadienyl rings, coining this molecule as “ferrocene”.4-6
Eventually, X-ray crystallography provided decisive evidence. In 1952, Fischer and Wolfgang Pfab published the first crystallographic data, showing the size and unit-cell dimensions which were consistent with the sandwich model.7 Subsequent studies confirmed this architecture, yielding bond lengths and thus fully establishing the three-dimensional structure. Chemists soon found that many metals could form similar sandwich complexes, and that the cyclopentadienyl rings could be replaced by other aromatic systems. This opened an entirely new field of organometallic chemistry. In 1973, the Nobel Prize was awarded to Fischer and Wilkinson for their pioneering work in the field.8,9
Ferrocene through the years
Ferrocene’s distinct properties forced chemists to rethink fundamental bonding theories. Its reversible one-electron oxidation to the ferrocenium cation (Fe³⁺ species) and the ease with which its Cp rings could be functionalised opened new avenues for chemistry. Researchers rapidly explored its redox chemistry, ligand substitution, and electrophilic and nucleophilic properties.4 These early investigations not only identified ferrocene as a versatile synthetic building block but also cemented the field of organometallic chemistry as a powerful discipline. The rapid growth of organometallic chemistry following ferrocene’s discovery is reflected in the increasing number of scientific publications encompassing ferrocene and its derivatives, especially from the 1960s onward. As shown in Fig. 2, annual publication output has risen significantly from the early years of research, demonstrating the compound’s continued relevance in all aspects of science including synthetic organometallic chemistry, catalysis, materials science, and medicinal chemistry, just to name a few.
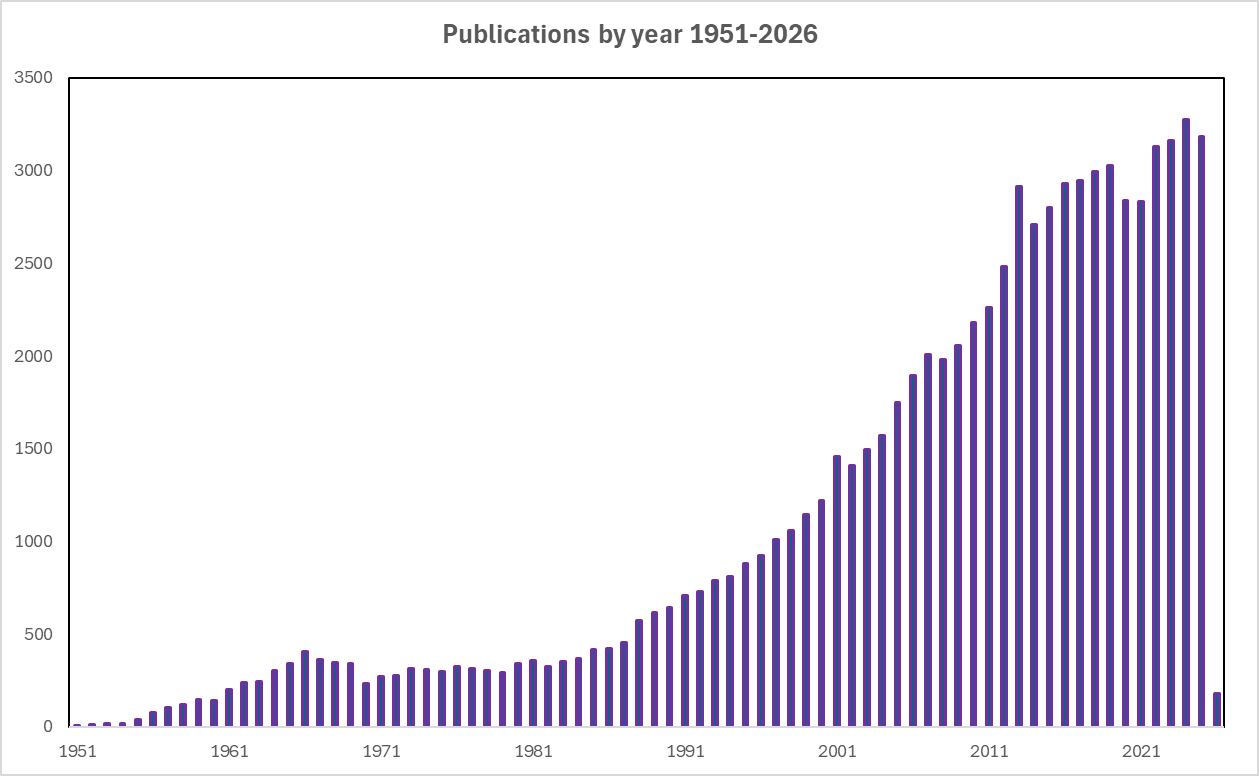
Fig. 2. Results from a SciFinder search of ferrocene and its derivatives, used to construct the graph of publications per year between 1951 and 2026.
Beyond traditional organometallic chemistry, ferrocene has played a pivotal role in the development of bioorganometallic chemistry, an interdisciplinary field that merges principles such as inorganic chemistry, biochemistry and medicinal chemistry. This field focuses on the use of organometallic complexes in biological and therapeutic contexts. Ferrocene’s distinctive properties makes it a compelling scaffold for drug design. Substituted ferrocenes, where one or both Cp rings are functionalised, can exhibit tuneable characteristics. The 18-electron configuration of the complex contributes to its extraordinary stability, and when oxidised to the ferrocenium ion, the compound can generate reactive oxygen species (ROS).10 As a result, ferrocene-based conjugates have shown promise against a range of diseases.
Ferrocene-based cancer therapeutics
Ferrocene-based complexes have been shown to exhibit anticancer activity, especially against hormone-dependent breast cancers. Tamoxifen, a potent cytostatic agent used to treat hormone receptor-positive breast cancers, is not without shortcomings. Long-term treatment can lead to acquired drug resistance, an increased risk of pulmonary embolism, and it has limited efficacy against hormone-independent breast cancers.11 To address these limitations, Jaouen and coworkers pioneered the design of ferrocifens, which are ferrocene-containing analogues of tamoxifen (Fig. 3). Ferrocifens are examples of molecules effective against both hormone-independent and hormone-dependent breast cancers, highlighting the potential of organometallic design motifs in medicinal chemistry. These ferrocene-tamoxifen hybrids displayed remarkable biological activity. The studies demonstrated that the hydroxy-substituted ferrocifens exhibit strong antiproliferative effects across a range of breast cancer cell lines including triple-negative breast cancer. The study revealed that certain ferrocifen derivatives performed comparably or even better than hydroxytamoxifen. Notably, ferrocene itself showed no antiproliferative activity; thus, the enhanced therapeutic effects of ferrocifens arise from the synergy between the ferrocenyl fragment and the tamoxifen pharmacophore.12-14 The activity of ferrocifen arises from its ability to undergo reversible oxidation from Fe(II) to Fe(III), a Fenton process. This redox cycling promotes the production of ROS which has damaging effects.15
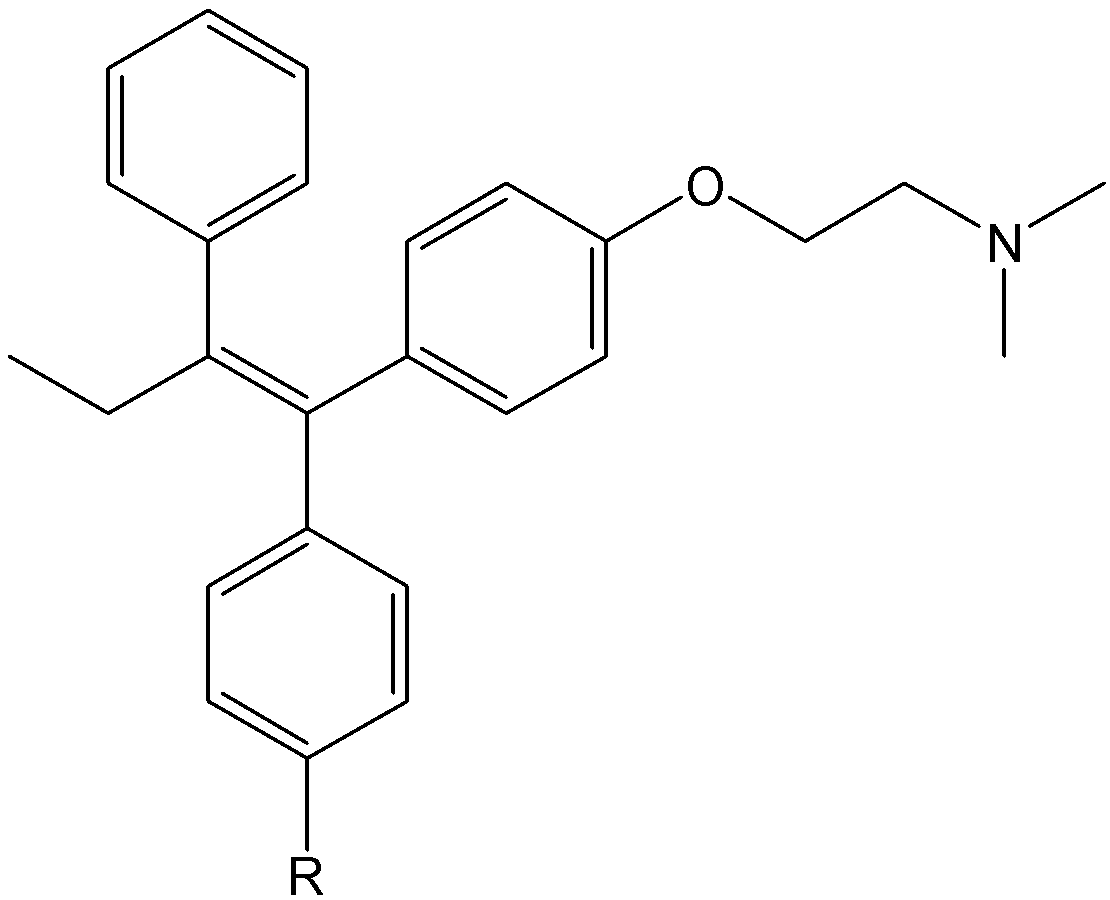
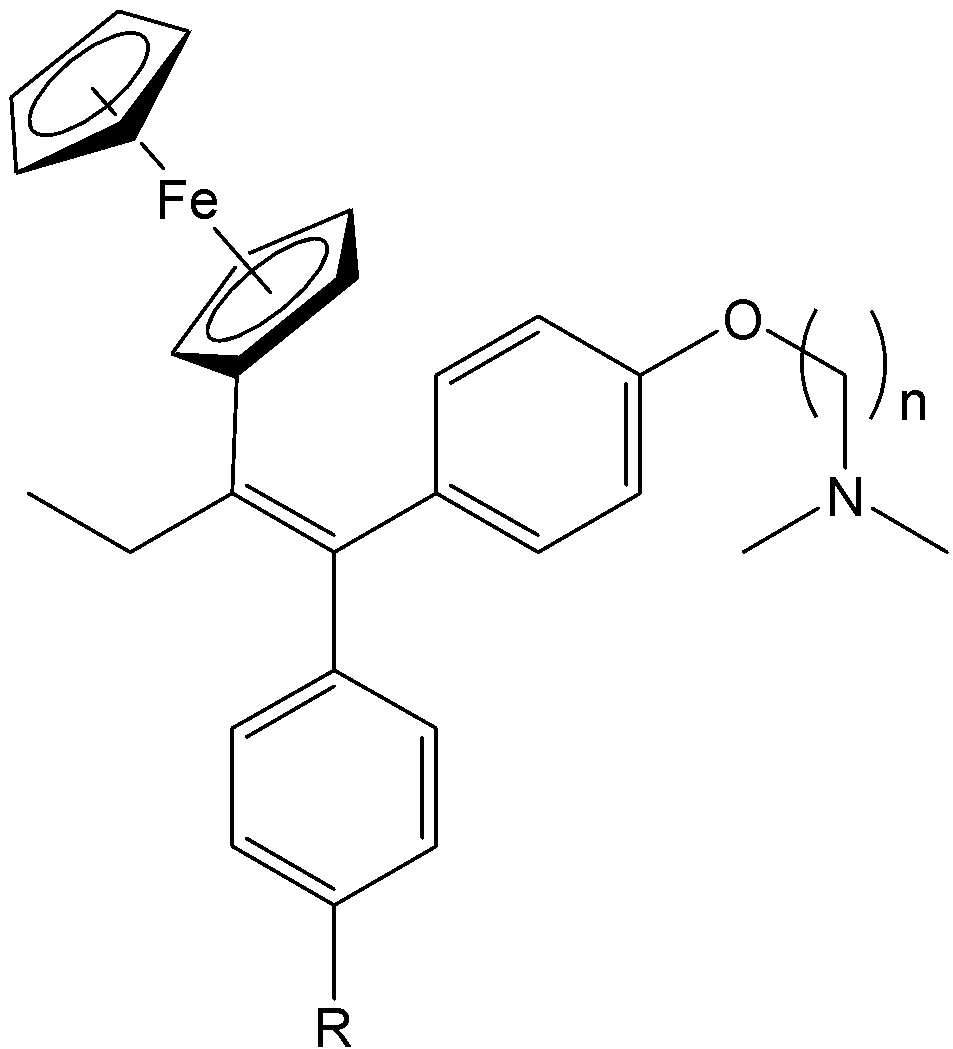
Fig. 3. Structures of tamoxifen (R = H), hydroxytamoxifen (R = OH) and ferrocifens.
Ferrocene in the fight against malaria
Ferrocene’s therapeutic potential extends beyond cancer. Malaria continues to represent one of the most serious parasitic diseases globally. In 2024, an estimated 282 million infections and 610,000 deaths were reported across 80 countries due to malaria. The burden was overwhelmingly concentrated in Africa, which accounted for 95% of all malaria cases (around 265 million) and malaria deaths (approximately 579,000) in the world. Tragically, children under the age of five were the most affected, representing about three‑quarters of all malaria‑related deaths in Africa.16 Malaria is caused by parasites of the genus Plasmodium and the parasite is spread by the female Anopheles mosquito. The major human-infecting species of Plasmodium, P. falciparum, P. vivax, P. ovale, and P. malariae, create substantial disease burden, with P. falciparum being the deadliest.
Traditional treatments such as chloroquine (CQ) (Fig. 4) mefloquine, and quinine have been highly effective; however, widespread drug resistance, especially in P. falciparum, poses a major challenge to malaria control efforts. The incorporation of ferrocene into antimalarial drug scaffolds has led to the development of promising new therapeutics. Among these, ferroquine (FQ) (Fig. 4), a ferrocene-chloroquine conjugate, stands out.17 Ferroquine has progressed through Phase IIb clinical development in partnership with Sanofi and Medicines for Malaria Venture (MMV) and continues to be explored in new combination regimens as it has been shown to overcome CQ resistance.18 FQ is significantly more potent than CQ, both against P. falciparum and in murine infection models. In some cases, ferroquine exhibits more than twenty-fold greater activity compared to chloroquine, with no major toxicity reported.19
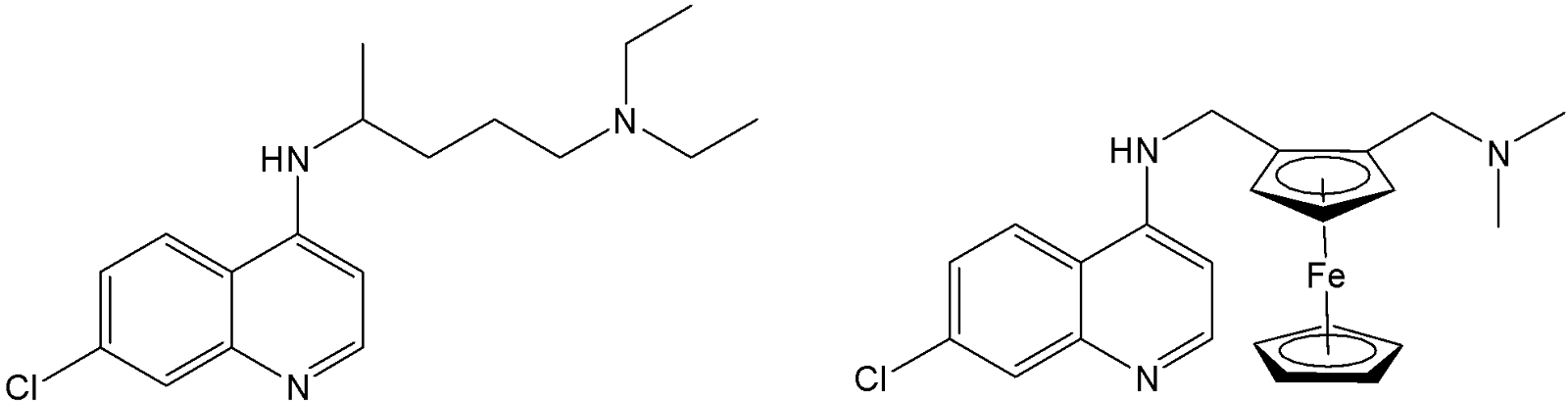
Fig. 4. Antimalarials chloroquine (CQ) and ferroquine (FQ).
Ferroquine works in various ways (Fig. 5). The intramolecular hydrogen bond in ferroquine plays a key role in its antimalarial potency. A strong internal hydrogen bond helps stabilise the molecule’s conformation, improving its ability to cross the parasite’s membrane into the digestive vacuole. The bond also increases the overall rigidity of the compound, supporting its activity against both chloroquine-sensitive and chloroquine-resistant strains of Plasmodium falciparum. Once inside the parasite, ferroquine is believed to disrupt the parasite’s ability to form insoluble hemozoin, a detoxification product of heme metabolism, thus interfering with its capacity to manage free heme, which is toxic to the parasite. Additionally, ferroquine in the presence of hydrogen peroxide (H2O2) generates hydroxyl radicals that causes damage to parasite proteins, which further enhances its antimalarial activity. Together, these effects contribute to ferroquine’s potency, particularly against CQ-resistant strains of the malaria parasite.20,21 Since ferroquine’s discovery, there has been a range of ferroquine analogues that have been prepared and their antimalarial activity tested. However, none of the complexes were found to be comparable to FQ. Other ferrocene-containing antimalarial agents have also been developed. Biot and coworkers synthesised ferrocenyl analogues of mefloquine and quinine, though these derivatives showed slightly lower antimalarial activity than their parent drugs.22
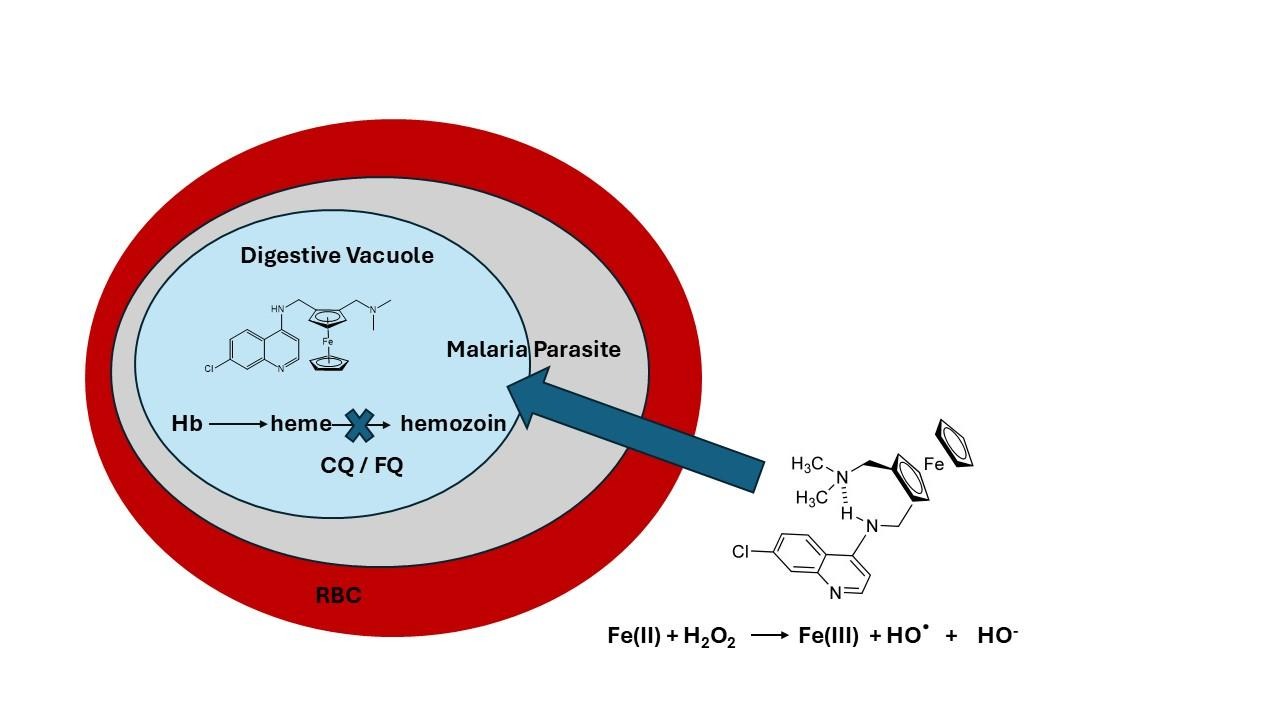
Fig. 5. Mechanism of action of ferroquine (FQ) in parasitised red blood cells (RBCs)
Overall, the ongoing interest in ferrocene arises from its rare combination of synthetic versatility, stability and redox activity. Whether in catalysis, molecular materials, sensing technologies, or therapeutic applications, ferrocene continues to inspire chemists across disciplines.23-25 Its discovery not only reshaped fundamental theories of chemical bonding but also catalysed innovations that continue to impact global health research today and will undoubtedly continue for the next 75 years.
References
- Ferrocene, Britannica, available at: https://www.britannica.com/science/ferrocene (accessed 29/01/2026).
- Kealy, T. J.; Pauson, P. L., Nature, 1951, 168, 1039–1040.
- Miller, S. A.; Tebboth, J. A.; Tremaine, J. F., J. Chem. Soc., 1952, 632–635.
- Wilkinson, G.; Rosenblum, M.; Whiting, M. C.; Woodward, R. B., J. Am. Chem. Soc., 1952, 74, 2125–2126.
- Fischer, E. O.; Pfab, W., Z. Naturforsch., B, 1952, 7, 377–379.
- Fischer, E. O.; Jira, R., J. Organomet. Chem., 2001, 637–639, 7–12.
- Takusagawa, F.; Koetzle, T. F., Acta Crystallogr., Sect. B, 1979, 35, 1074–1081.
- Nobel Prize in Chemistry 1973, available at: https://www.nobelprize.org/prizes/chemistry/1973/summary/ (accessed 29/01/2026).
- Seyferth, D.; Davison, A., Science, 1973, 182, 699–701.
- Štěpnička, P., Dalton Trans., 2022, 51, 8085–8102.
- Cuzick, J.; Powles, T.; Veronesi, U.; Forbes, J.; Edwards, R.; Ashley, S.; Boyle, P., Lancet, 2003, 361, 296–300.
- Top, S.; Tang, J.; Vessières, A.; Carrez, D.; Provot, C.; Jaouen, G., Chem. Commun., 1996, 955–956.
- Top, S.; Dauer, B.; Vaisserman, J.; Jaouen, G., J. Organomet. Chem., 1997, 541, 355–361.
- Jaouen, G.; Top, S.; Vessières, A.; Leclercq, G.; Quivy, J.; Jin, L.; Croisy, A., C. R. Acad. Sci., Ser. IIc, 2000, 3, 89–93.
- Osella, D.; Mahboobi, H.; Colangelo, D.; Cavigiolio, G.; Vessières, A.; Jaouen, G., Inorg. Chim. Acta, 2005, 358, 1993–1998.
- World Health Organization, World Malaria Report 2025, available at: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2025 (accessed 29/01/2026).
- Biot, C.; Glorian, G.; Maciejewski, L. A.; Brocard, J. S.; Domarle, O.; Blampain, G.; Millet, P.; Georges, A. J.; Abessolo, H.; Dive, D.; Lebibi, J., J. Med. Chem., 1997, 40, 3715–3718.
- Medicines for Malaria Venture (MMV), Ferroquine programme update, available at: https://www.mmv.org/newsroom/news-resources-search/artefenomelferroquine (accessed 29/01/ 2026).
- Delhaes, L.; Abessolo, H.; Biot, C.; Berry, L.; Delcourt, P.; Maciejewski, L. A.; Brocard, J. S.; Camus, D.; Dive, D., Parasitol. Res., 2001, 87, 239–244.
- Dubar, F.; Egan, T. J.; Pradines, B.; Kuter, D.; Ncokazi, K. K.; Forge, D.; Paul, J.-F.; Pierrot, C.; Kalamou, H.; Khalife, J.; et al., ACS Chem. Biol., 2011, 6, 275–287.21. Dive, D.; Biot, C., Curr. Top. Med. Chem., 2014, 14, 1684–1692.
- Dive, D.; Biot, C., Curr. Top. Med. Chem., 2014, 14, 1684–1692.
- Ornelas, C.; Astruc, D., Pharmaceutics,2023, 15, 2044.
- Stringer, T.; Wiesner, L.; Smith, G.S., Eur. J. Med. Chem., 2019, 179, 78–83.
- Hofmann, B. J.; Aljohani, E. T.; Cicovacki, N.; Lee, I.; Warren, D. T.; Sobolewski, A.; Stringer, T.; Lord, R.M., ChemBioChem, 2025, 26, e202400759.
- Tremlett, W. D.J.; Crowley, J. D.; Wright, L. J.; Hartinger, C. G., Dalton Trans., 2024, 53, 14742–14751.





